


Water treatment in today’s modern world requires highly technical, high performance separation products to achieve increasingly stringent treated water qualities, or to provide the lowest cost of water production. However, more than one separation technology is often applied to achieve the demanded quality. This development was a main consideration for LANXESS to start production of Lewabrane® reverse osmosis (RO) membrane and elements at a new production site in Bitterfeld, Germany.
Reverse osmosis membranes
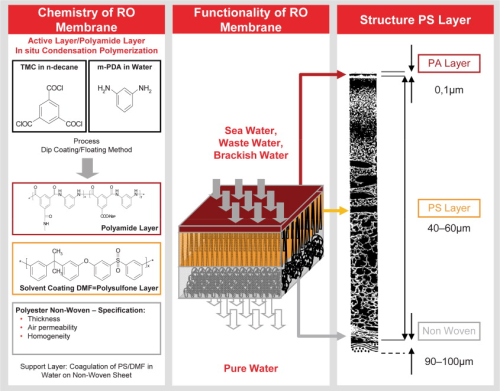
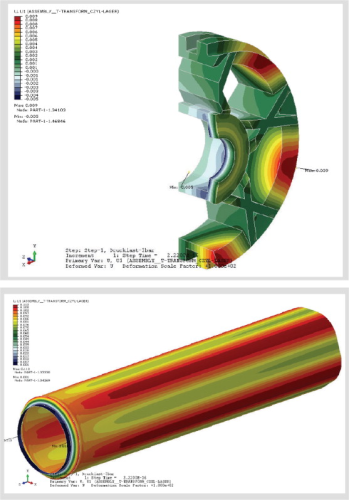
The dominant reverse osmosis membrane structure is based on a thin film composite membrane (see Figure 1). The barrier (or rejection) layer is a 0.1μm thick polyamide layer which is supported by a polysulfone substructure. The polyamide layer is formed by a polymerisation process. Although thin film composite membranes based on this process have been used for more than thirty years, the newest technology now offers the possibility to control the polymerisation process more precisely. Accordingly, a strong focus of our membrane development was the enhanced polymerisation degree of the polyamide layer. A higher polymerisation degree improves the mechanical and chemical stability of the thin barrier layer offering greater durability. Additionally, the negative charge on the membrane surface is reduced which leads to a lower cationic adsorption (fouling) on the membrane surface. Due to its chemistry, the surface of a polyamide membrane is usually negatively charged, and often results in cationic fouling that is extremely difficult to remove. A typical example of cationic fouling is fouling with iron. Iron chloride (FeCl3) is a very common flocculation chemical used in pre-treatment systems. If the dosing is too high, even just for a short period, the cationic fouling can irreversibly foul the RO membrane surface. Apart from a well adjusted iron chloride dosing system, a lower negative surface charge is the best option to reduce the fouling potential of this event. The membrane separation composite layer is the most essential part of the RO separation process. Before use, this critical component is assembled into a device, called an RO element. The winding process for spiral wound RO elements involves many steps all of which need to be carefully controlled. The LANXESS manufacturing process involves state-of-the-art robotic equipment to prepare the RO element carefully to exacting mechanical specifications. Much of this development was conducted with the assistance of outside Institutes that applied modern computer aided design capabilities to confirm mechanical strength and optimise hydrodynamic design (see Figure 2). This kind of critical development process was necessary to assemble the improved membrane chemistry into a modern RO element.
Field testing
The first field tests of the new Lewabrane® RO B400HR elements started in January 2012. The elements were placed in an existing RO plant, which is treating 40m3/hr of Rhine river water after an ultrafiltration system. A total of six elements were installed in a pressure vessel. The RO system also contained several pressure vessels with RO elements from another supplier. These elements were installed approximately one year previously, and were operated in parallel to the Lewabrane® pressure vessel. The entire RO system was operated in a two stage system with a 6:3 array using six element pressure vessels. In comparison with the installed elements from another supplier, it could be shown that the Lewabrane® elements provided a flux value of the same order of magnitude. The TOC and silica rejection were measured periodically during the field testing, while the TOC rejection was measured at approximately 95-96%, the rejection of the total silica was measured at around 99.3%. In conclusion, it was demonstrated that the new Lewabrane® RO membrane element performs in a similar manner under the same operating conditions.
| Process | Demineralisation | Desalination |
| Limits | Conductivity < 2 μS/cm TOC < 500 ppb SiO2 < 50 ppb | Conductivity < 0.055 μS/cm TOC < 100 ppb SiO2 < 10 ppb |
| Technology | Ion Exchange Reverse Osmosis Electrodialysis | Ion Exchange (mixed bed) Electrodeionisation (EDI) |
Ion exchange
The reason that LANXESS undertook the step to add RO membrane technology to its separation products portfolio is that reverse osmosis is a complementary technology to ion exchange (IX) resins. LANXESS has produced ion exchange resins for more than 70 years under the brand name Lewatit®. As a general rule, reverse osmosis can efficiently desalinate water with a high salinity, but ion exchange can selectively remove certain ions from the water. Table 1 shows which separation technique could be used, depending on the requested permeate quality. In modern separation applications, combined RO and IX processes are not only used in applications like the desalination of boiler feed water, but in other process applications like the removal of boron from seawater, or the treatment of produced water from unconventional gas resources. These applications have recently been under much discussion because of increasing public concern for water quality and environmental considerations. The production of gas from unconventional sources is growing rapidly worldwide. Water that comes up along with the gas is not only challenging to treat, but must often meet strict regulatory standards prior to release into the environment. Such regulations pose challenges to water treatment system designers. While varying significantly, typical waters may have a TDS range of 2,500–10,000 mg/l, total alkalinity of 1,000–3,000 ppm as CaCO3, and a pH in the order of 8-9. Since most inland areas are sensitive to the discharge of salts, the concentrate from a water treatment process will require specialist disposal. Therefore, minimising the volume of waste concentrate is paramount to the success of the application. RO processes with a high recovery rate and low usage of chemicals are one solution. To achieve this goal, softening with IX as pre-treatment and inter-stage treatment for a three stage, single pass RO unit is one option.
Softening
In order to achieve a high recovery, softening down to ppb levels of hardness is required. To achieve this reliably, particularly for high salinity water, a selective IX process is used. One example of this is the weak acid cation resin type (for example, Lewatit® CNP80), which is typically used prior to the RO treatment for brackish waters. Another example is chelating resins, which are able to soften effectively to ppb levels, even from saturated brine solutions. This type is typically used to soften the concentrate from an RO plant, ahead of further RO treatment. An iminodiacetic acid chelating resin (for example, Lewatit® MonoPlus TP208) is typically selected when strontium and barium removal is important; otherwise an aminophosphonic acid resin (for example, Lewatit® MonoPlus TP260) is preferentially used. In most applications a chemical treatment with acids or antiscalant is used as pre-treatment for an RO process. The softening process with IX has advantages if, like in the example described, the discharge of salts is difficult, or if the solubility products (Ksp) of the salts are far above the saturation limit so that an antiscalant cannot be used for the application (see Table 2).
| Scale forming compound | Conservative saturation level |
| CaSO4 | 230% |
| BaSO4 | 6,000% |
| SrSO4 | 800% |
| SiO2 | 150% (or 200ppm) |
| CaCO3 | LSI > 1.8, SDSI >1.0 |
Boron removal
In contrast to the produced water treatment example described IX is used as a post-treatment for boron removal. Boron removal with RO alone is done at pH 9. At this pH, the boron is partially negatively charged and the rejection can be up to 90% with seawater RO, and 75% with brackish water RO elements. To achieve a limit of below 0.5 mg/l boron in the permeate, an additional RO treatment of the first permeate is necessary (by a two pass or partial two pass system). The pH adjustment is done in front of the second pass in such a case. An alternative for this process is a post treatment with IX. Although only a few plants are installed with this technique, this process has some clear advantages if the customer equests a low boron concentration (0.3 mg/litre). In a pilot test of a seawater desalination plant the boron level could be reduced from 0.7 mg/l (after RO) to 0.2 mg/l (operating capacity of 2.6 g/l). Since IX is a separation process with a high selectivity, mainly boron is removed and the capacity of the resin is not exhausted by other ions. Similar processes can be used to remove other critical compounds selectively after an RO process, like arsenic or heavy metals. Most engineers use customised software from product manufacturers to design ion exchange or reverse osmosis systems. Since July, a new comprehensive software program, designated LewaPlus, is available from LANXESS. This software can calculate reverse osmosis and ion exchange arrays inside the same program. As a consequence, water treatment plants with a two pass system or hybrid processes using RO followed by IX can be compared, allowing the designer to quickly optimise the water treatment plant. Additionally, the effects of process variables, like temperature, can be calculated and assessed for a whole system. When designing an RO plant, the software offers the possibility to get a recommended array based on the information that has been entered. Further updates are planned this year, for example the option for post-treatment with IX, and a detailed cost and energy calculation for the RO design. In making an economic validation of RO and IX processes, the cost of discharging the concentrate is often important. However, the salt concentration of the feed is usually of primary interest. While the specific costs for demineralised water using IX is dependent on the salt concentration of the feed water, the specific cost for an RO plant is constant for a broad range of salt concentrations. On the other hand the specific costs of the RO treated water are starting at a higher level so that the breakeven point (intersection) shows the designer where the salinity value of IX and RO have the same costs. Apart from the economic validation other reasons may appear as to why a RO or IX is selected for a process. In general, RO process is preferred if easy handling is a critical selection issue while IX is preferred if a high selectivity is beneficial.
Conclusions
LANXESS believes that both IX and RO technologies will continue to grow strongly in the near future. Seawater desalination is rapidly growing with an expected growth rate of 12%; brackish water at a slightly lower growth rate. The RO membrane process clearly has a bright future. In addition, with water treatment processes demanding greater efficiency and selectively, IX also has a bright future. The modern technology of water treatment requires the combination of several technologies. For example, the integration of different membrane processes, such as ultrafiltration and RO membrane separation, or the combination of several techniques like RO and IX, or RO and EDI. LANXESS now offers two state-of-the-art process solutions (RO and IX) to allow the process designer to optimise the water treatment process with the goal of a lower cost and higher reliability of water treatment for the user.




